 Courtesy of cancer.gov
Courtesy of cancer.gov
This year's gathering of the American Society of Clinical Oncology (ASCO) in Chicago will be an informative one. Cancer research and treatment is at the forefront of a tremendous and exciting frontier.
For a very long time the three main modalities of cancer treatment have been surgery, chemotherapy and radiation. But thanks to recent advances we can add immunotherapy into the mix, what some call “the fourth pillar” of cancer treatment. As research progresses, the potential drug treatments being churned out are actual game changers, which means that the five-year survival benchmark could become a thing of the past.
Immunotherapy is also known as "targeted therapy" where drugs specifically target molecules on cancer cells that are responsible for their growth and spread. They do not have the broad spectrum of action that chemotherapy does, and whereas chemotherapy kills cancer cells (in addition to normal cells) targeted therapies will block the tumor cells from proliferating.
Targeted therapies are focused on the aberration present in the cancer, whether that is abnormal proteins on the cell surface or chromosomal abnormalities within the cell nucleus. Targeted therapies can be monoclonal antibodies, which are large molecules that interact with targets outside the cell, or they can be small-molecule compounds that are capable of entering the cell.
One promising immunotherapy is what is called "checkpoint inhibitor." Checkpoints are present on immune cells as way to regulate their activity – to be able to attack only foreign antigens and spare surrounding tissue from collateral damage. Checkpoints also inhibit autoimmunity. Tumors have been able to manipulate this feature for their benefit so they can resist immune recognition and attack – specifically the T-cells of the immune system which are the immune responders to tumor cells.
Keytruda, a checkpoint inhibitor, has received a significant amount of media attention considering the success it has had in treating ex-president Jimmy Carter’s aggressive melanoma. In conjunction with surgery, radiation and multiple doses of this drug, Carter said all signs of the cancer were gone. It has also received attention because it costs about $150,000 per patient per year.
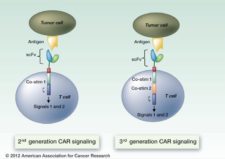
Checkpoint inhibitors are not the only medications making waves, however. Also in the pipeline is what is called ‘"chimeric antigen receptor T-cells" or CARTs. This treatment engineers a patient’s own immune cells to recognize and attack their own tumors. The modified immune cells are then re-infused back into the patient in an approach called "adoptive cell transfer" (ACT). It is like “giving patients a living drug,” according to Renier J. Brentjens, MD, Ph.D, of Memorial Sloan Kettering Cancer Center (MSKCC) in New York, also one of the premier researchers in CART therapy.
A study, published in the New England Journal of Medicine, revealed tremendous success using CART in pediatric patients with acute lymphoblastic leukemia (ALL) who either relapsed or were unresponsive to treatment. Leukemias are the most common pediatric cancer and ALL is the predominant pediatric leukemia. Of the 30 children enrolled in the study, complete remission was achieved in 27 patients (90 percent) — even among those patients for whom stem-cell transplantation had failed. The drawback was that a severe and overwhelming immune response called a "cytokine storm," which is potentially fatal, was seen in 27 percent of patients.
“All the preliminary clinical data that have been generated, both by us and by our competitors indicate that the treatment benefit will be transformative,” stated David Chang, MD, Ph.D, chief medical officer of Kite Pharma Inc., who will present more results from CART trials at this year's ASCO meeting.
With the successes of these immunotherapies, the field has been opened up to the possibility of successfully treating other cancers thought to be largely considered death sentences, such as pancreatic and brain cancers. The White House was right in calling these recent advents an "inflection point" in cancer science with the real possibility of big gains.



